
Center for Negative Carbon Emissions
We are advancing carbon management technologies that can capture carbon dioxide (CO2) directly from ambient air in an outdoor operating environment.
Arizona State University’s Center for Negative Carbon Emissions (CNCE), with our commercial partner, Carbon Collect, is testing a prototype technology that would remove CO2 from the air through the use of MechanicalTrees™.
Widespread use of such a technology could help draw CO2 out of the air and reduce global warming.
The CO2 collected by the Mechanical Trees ™ can then be converted into a carbon neutral fuel or other useful chemicals, or disposed of to cancel out present or past emissions. The specific design licensed to Carbon Collect is an ASU innovation developed through Salt River Project sponsorship. ASU and Carbon Collect are partnered on research and commercialization of
the Mechanical Tree ™ design.
On the lab scale, we can demonstrate that various sorbent varieties and regeneration techniques can increase the concentration of CO2 from the amount that is in ambient air, 400 parts per million (ppm), or 0.04%, to an enriched stream of up to five percent (then compressing to the desired end-use). This passive process does not blow air but relies on the wind. Specific projects to demonstrate these processes are listed below.
World-renowned researchers

Matthew Green
Associate Professor
Green’s research focuses on the design and synthesis of novel, ion-containing polymers to be used in applications such as water purification, carbon dioxide capture, nano composites, and micellar solution assemblies.

Klaus Lackner
Center Director and Professor
Klaus Lackner is the director of CNCE and a professor in the School of Sustainable Engineering and the Built Environment of the Ira A. Fulton Schools of Engineering at ASU.
Innovative research projects
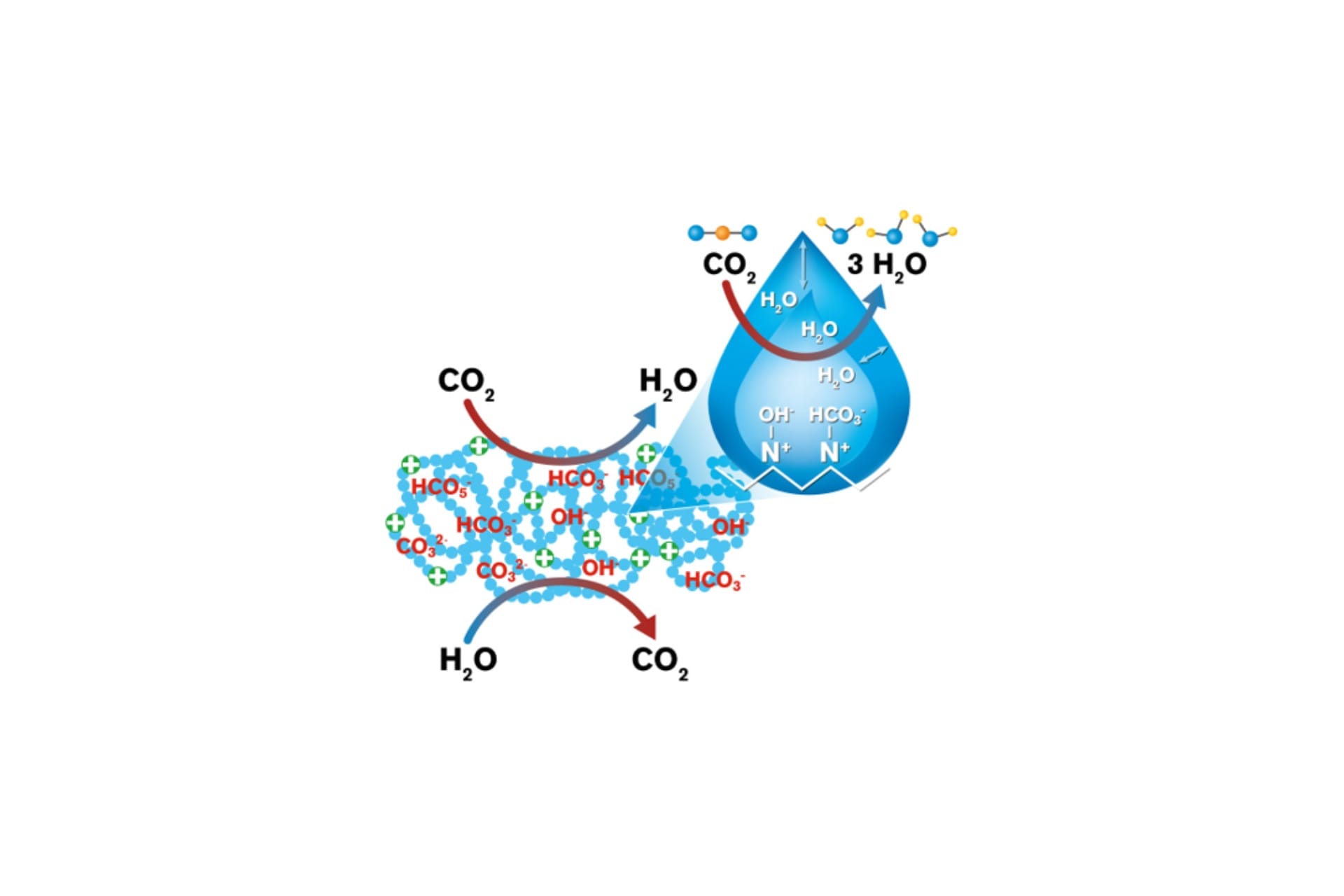
Molecular mechanisms of moisture-driven DAC (MissionDAC)
MissionDAC unites a diverse team from Northern Arizona University (NAU), ASU and University of Texas at Austin to engage in coordinated fundamental research of CO2 sorbent materials that release captured CO2 using moisture (water) rather than energy intensive heat or vacuum pressure.

AUDACity
ASU’s DAC polymer-enhanced cyanobacterial bioproductivity (AUDACity) project focuses on developing innovative sorbent materials that capture carbon dioxide (CO2) from the atmosphere when dry and deliver it when the material comes into contact with cultivation liquids, thereby enhancing the productivity of cyanobacteria.

MechanicalTreeTM
The Mechanical Tree™ is a groundbreaking carbon dioxide (CO2) direct air capture (DAC) technology and a pioneering advancement to address climate change. The first Mechanical Tree ™ in the world was installed by Carbon Collect at ASU, which will enable outdoor, pilot-scale DAC research at ASU CNCE. Natural air flow from wind brings air in contact with specialized materials (sorbents) that remove CO2 without the need for fans or blowers.” to make it even with the other text lengths.
CNCE considers the economic, political, social and environmental ramifications that will arise with the availability of an affordable air capture technology.
The center’s long-term goal is to become the intellectual leader in this new field of sustainable energy infrastructure design, which is critical to achieving a carbon negative energy economy.
